The right solution for every challenge
Do you want to know which applications can benefit from our technologies and flexible production platforms? Would you like to see specific examples of the wide range of complex projects we have realized around the world? Are you interested in learning how our customers experience the collaboration with Harro Höfliger? You will find out all of this and much more right here:
- Experience reports – customer projects around the world
- Product examples – applications in a variety of markets
- Services – the multiple facets of our services
- Machines – features and performances in detail
- Innovations – unique new solutions
Stories

- All
- Aseptic Processes
- Capsule Filling & Weighing
- Customized Products & Solutions
- Diagnostic Products
- Eye Care
- Inhalation Products
- Medical & Pharmaceutical Device Assembly
- Microneedles
- New Drug Delivery Systems
- Ostomy & Continence Care
- Packaging
- Portion Packs
- Services
- Transdermal & Oral Delivery Systems
- Wound & Skin Care
Technology platform for the production of lipid nanoparticles
Nanoparticle formulations can improve drug stability, increase their bioavailability and deliver them to their target site. The new technology platform, developed by leon-nanodrugs, enables fast, efficient and flexible GMP manufacturing of nano encapsulated products.
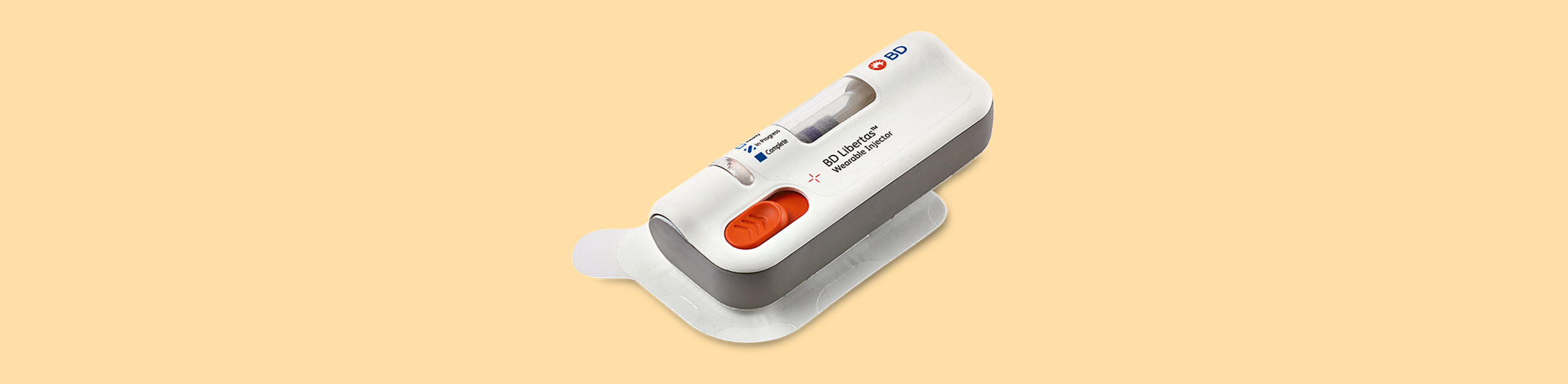
Large volume Subcutaneous self-administered injection by means of a Wearable Injector
BD Libertas™ Wearable Injector is a drug delivery solution designed for the treatment of chronic diseases requiring large volume (2-5 ml) subcutaneous biologic administration. Harro Höfliger designed an assembly system for test and clinical trial quantity production.
Re-usable Inhaler Respimat®
Since 2004, the Respimat® has been ensuring easy inhalation for patients with diseases such as asthma and COPD. In 2019, Boehringer Ingelheim launched the re-usable version of this inhaler.
Implant syringes for more safety
The Gaplast implant syringe offers protection and safety for patients. It is used, for example, in the treatment of oncological diseases.
Capsule production for dietary supplements
The demand for dietary supplements reached a new peak last year. For the filling of capsules, Warnke - one of the market leaders in this field - has been relying on machine technology from Harro Höfliger for many years.
Protection against needlestick injuries
The Safe’n’Sound® device from Nemera reliably prevents needlestick injuries. Its robust and ergonomic design makes it suitable for both naïve and experienced users.
One machine, many capsule products
Every now and then, the Argentine pharmaceutical company Gador fills its capsules with extraordinary content – for example with tablets. For this and many other products, Harro Höfliger designed the appropriate dosing stations.
The Papillon: A simple Dry Powder Inhaler
With the aim of helping pharmaceutical companies enter the market more quickly, Hovione Technology has developed the Papillon dry powder inhaler. Thanks to its simple design, it is extremely easy for patients to use.
Personalized medicine from a pharma printer
Printed medicines tailored to each patient's individual needs: That is what DiHeSys (Digital Health Systems) stands for. Harro Höfliger as their partner provides support in the design of the pharmaceutical printers required for this purpose.
Sustainable and safe packaging of vials
Saving resources and avoiding waste: On a new line from Harro Höfliger, Recipharm packages vials without plastic.
Preventive maintenance for diagnostic turnkey line
A Preventive Maintenance Plan will facilitate the maintenance of a complex machine system from Roche in the future. With its support, technical malfunctions should be avoided, and production output increased.
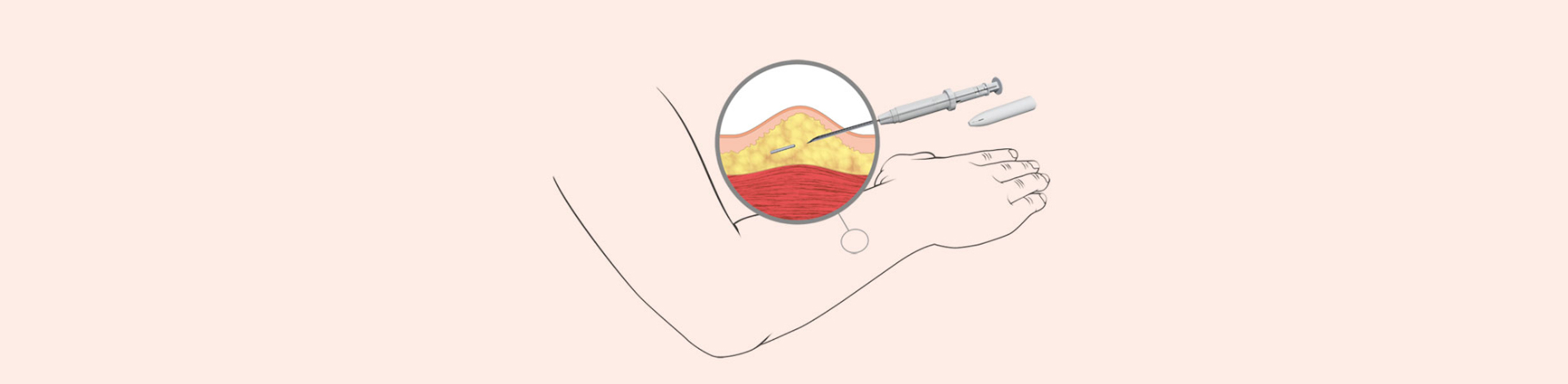
Microneedle patches to tackle migraine
Zosano Pharma has developed an intracutaneous microneedle system that helps active ingredients enter the bloodstream more quickly. One example of its application is the treatment of migraine.
Semi-automated filling of a dry powder inhaler
The start-up PureIMS develops and produces dry powder inhalers (DPIs) based on the Cyclops™ platform. Harro Höfliger provided support for the scalability and automation of the processes.
Future technology: Water sealing of pouches
Water-soluble pouches, also known as pods or tabs, consist of PVOH (polyvinyl alcohol) film filled with detergent or cleaning agent. The pre-dosed products offer the consumer a convenient application, which is especially safe thanks to water sealing.
Side-Loading for Top-Opening Packaging
Packaging of products in an attractive, sustainable and flexible way: Side-loading is a convenient solution for the safe, plastic-free packaging of products in top-opening cartons.
High-performance production of a special capsule solution
With the Accordion Pill®, Intec Pharma has developed an innovative capsule platform that provides continuous drug release. The manufacturing technology for the product was supplied by Harro Höfliger.
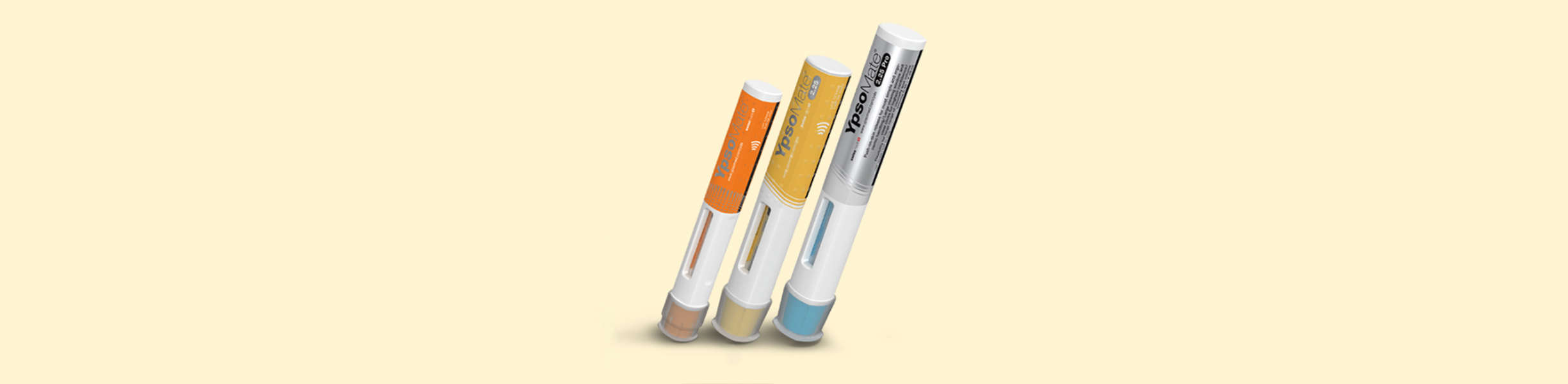
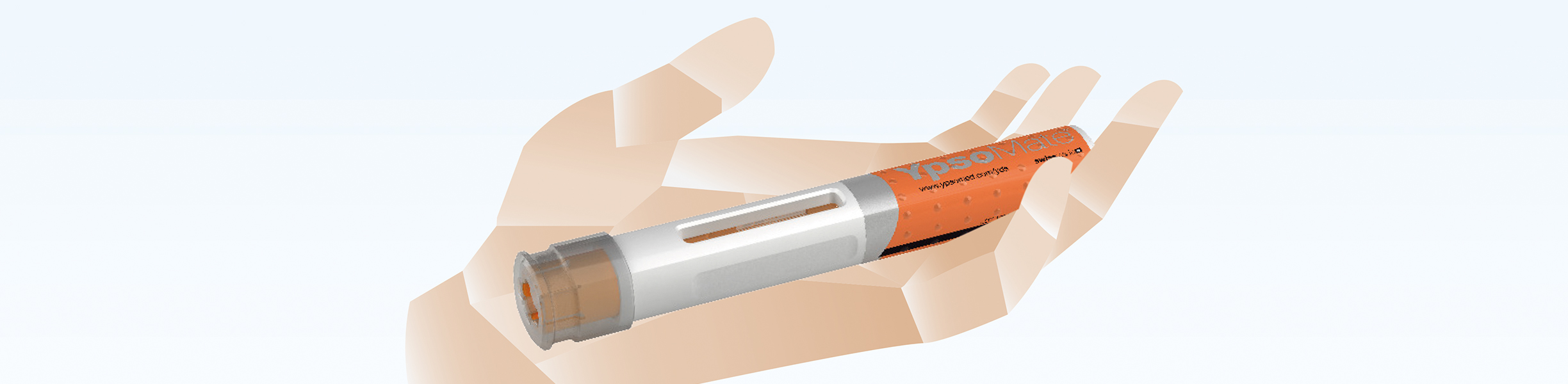
Flexible final assembly of auto-injectors
Ypsomed has developed an auto-injector product family for a wide range of applications. The Harro Höfliger Assembly Lab machine provides a flexible final assembly of the YpsoMate® auto-injectors.
Subcutaneous implants as a drug depot
Subcutaneous implants cover a wide range of applications. In the future, they can replace tablets and syringes.
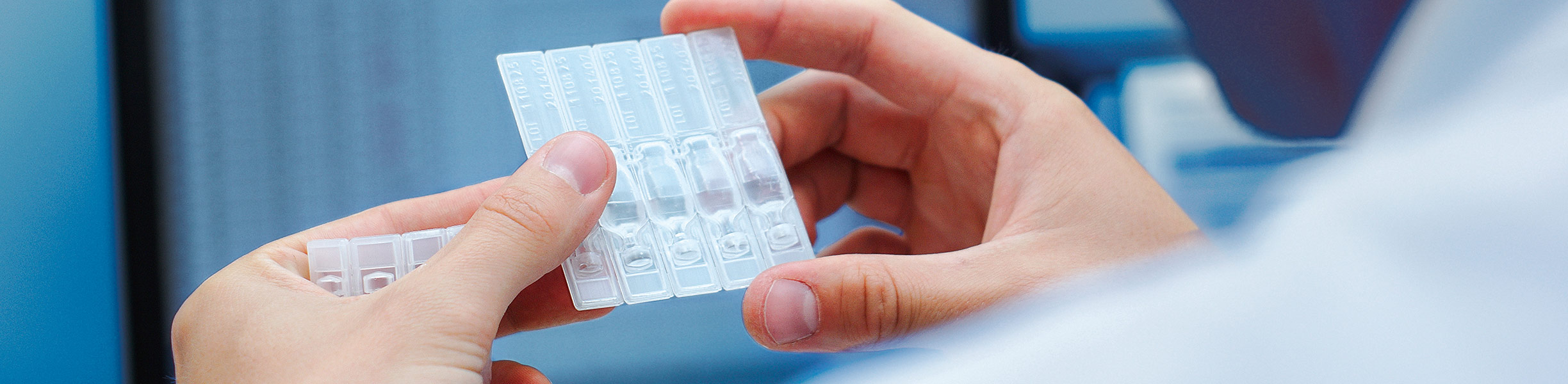
ScreenTape: Reliable quality assurance for genetic testing
For a rapid quality control of DNA and RNA samples before expensive testing, the Screen Tape diagnostic kit was developed. From the beginning, Agilent has relied upon the technologies from Harro Höfliger.
Individual filling solutions for powder
If powders cannot initially be dosed due to their particle shape, size, stability or density, new and creative solutions have to be found.
Efficient packaging of syringe sets
The demand for an oncological drug is increasing. AstraZeneca has therefore decided to automate the packaging processes with a turnkey line.
Microneedle patches facilitate vaccination protection
Microarray patches are thought to be a future alternative to conventional vaccination campaigns. VAXXAS and Harro Höfliger have been working on an affordable and scalable implementation.
Allergy diagnostics with the Immunocap™ Test Dispenser
For decades, the ImmunoCAP™ test dispenser by Phadia AB, a Thermo Fisher Scientific company, has been used to support the diagnosis of allergies.
Aseptic filling of eye drops
Working at computer screens, air conditioning systems and artificial light – the demand for eye drops is increasing. Rommelag CMO ensures your filling process in BFS-container.
Medical cannabis in exact doses
Founded in 2011, Syqe Medical from Tel Aviv is developing medical dosage forms for cannabis and other medicinal plants.
Auto-injectors and injection pens reliably produced
The Swiss device manufacturer Ypsomed and Harro Höfliger have established a manufacturing network
Safety inhalation with the NEXThaler
Inhalable drugs provide relief to those affected by asthma and COPD worldwide. With NEXThaler®, the Italian company Chiesi offers a simple and safe inhalation device.
Sterile filling of infusion bags
New B Innovation Limited (New ß Innovation) uses a partially automated aseptic filling and sealing line for pre-sterilized infusion bags.
Filling quantity control of inhalation blisters
X-ray technology instead of destructive random sampling: The X-ray module enables the inline verification of powder filled, sealed aluminium blister strips for Dry Powder Inhalers (DPI).
Containment concept for capsule filling machines
Harro Höfliger has designed a compact system for its capsule filling machines of the Modu-C family, which offers reliable protection for the operator, as well as the environment.
Respimat®: Inhaler for liquid formulations
Respimat® from Boehringer Ingelheim is a successful product for the treatment of lung diseases such as Asthma and COPD. Harro Höfliger and partner companies from Excellence United are involved in the filling and packaging.
Production of water-soluble pouches
Portion packs for dishwashers and washing machines are the trend. They are easy to handle and avoid overdosing. How are they manufactured with the desired shape and fill quantity?
High-speed line for TTS/TDS products
When it comes to the production of patches for the treatment of asthma and pain Nipro Patch relies on Harro Höfliger’s many years of expertise in web converting as well as in transdermal systems.
Product consulting by Pharma Services
Accelerating processes, and minimizing project costs and risks – this is what the Harro Höfliger Pharma Services experts specialize in.
Plastic applicator for administering a nasal gel
A new single-use applicator ensures the precise and hygienic application of a thixotropic gel, which encapsulates harmful particulates and reduces the toxic risk.
Medication intake through oral and buccal film strips
Oral (ODF) and buccal (MBF) film strips offer huge potential for the pharmaceutical market. Harro Höfliger has a well-established machine program for the production and packaging of film strips.
Navel patch heals abdominal pain
The Shanxi Yabao Pharmaceutical Group produces homeopathic patches for the Chinese market. The “gingerbread” patch is manufactured on a Packing and Four-Side Sealing Machine from Harro Höfliger.
XStraw: dosage form for pediatrics and geriatrics
XStraw® is the name of a new dosage form using a drinking straw. The straw is pre-filled with a drug and enables a more comfortable intake for children and patients having diffculty swallowing.
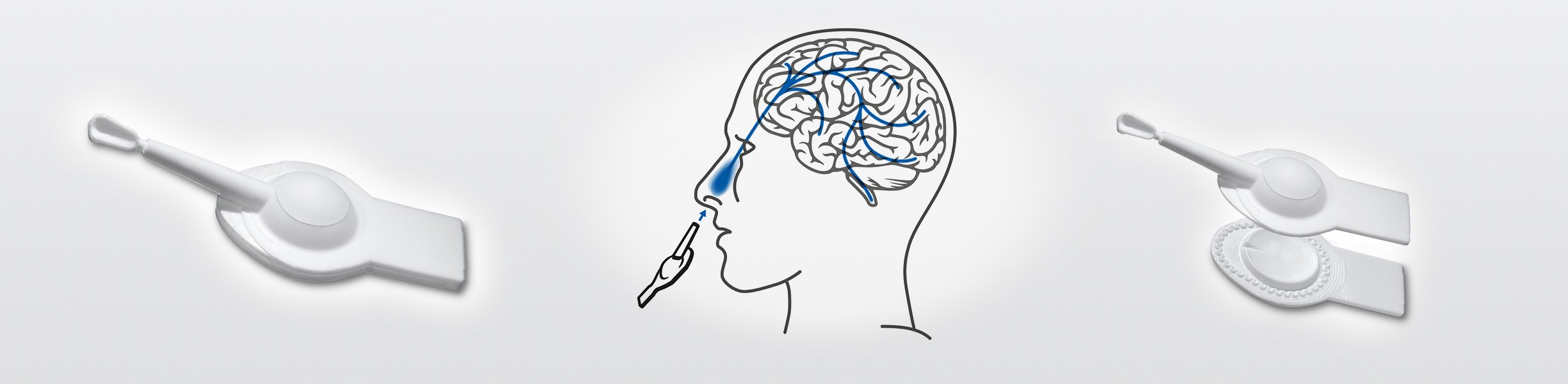
Nasal administration of molecules active in the brain
M et P Pharma AG found in Harro Höfliger a skilled partner for the filling and sealing their nasal devices.
Labelling and packaging of eye drops
Harro Höfliger produced a labeling and packaging line for Pharma Stulln – a GMP-compliant production line – within a very tight deadline. The limited space available required special line planning.
Lab-on-a-chip production for rapid tests
So-called mini labs make it possible to analyze samples at the “Point-of-Care”. Harro Höfliger supplied a filling and sealing station for the production of test elements at Hahn-Schickard in Freiburg.
Smart patches equipped with on board technology
Mobile medical monitoring by means of a smart device: Dressings with small sensors and wireless technology are comfortable to wear and accurately record patients' data.
Gentle handling of lyobead pellets
For the handling of lyophilisates, Harro Höfliger offers a unique automated solution – the Lyo Bead Dispenser. The freeze-dried pellets are safely conveyed and accurately dosed via air flow.
Administration of insulin using DPI inhalers
Powder inhalers help diabetics: Harro Höfliger designed and built two filling and assembly systems for dry-powder inhalation systems for the US based MannKind Corporation.
Delta Pouch: Packaging of powdered products
The delta-shaped foil packs each contain a single portion and can be opened quickly and easily.
Secure cartoning of double bags
Klocke Pharma-Service and Harro Höfliger have worked together for more than 25 years. A few months ago, they implemented the third line for packaging double bags, based on a MKL horizontal cartoner.